
Figure 1 – 2D fITI System prototype.

Figure 2 – 3D fITI System prototype.




Functional Infrared Thermal Imaging based on the spectral analysis of the skin microvascular blood flow to investigate the role of perfusion components in pain sensation as promising biosignals and biomarkers for PAIN assessment: towards a novel and objective instrumental technique for pain measurement
Funding Programme: PRIN 2022 Call “PRIN 2022 DD n. 104 02-02-2022”
CUP: C53D23001540006 – Project Code: 2022R2KZ7B
Project Duration: 28/09/2023 – 28/02/2026
Proposing Research Team




Rosario Morello was born in Reggio Calabria, Italy, in 1978. He received the M.Sc. Degree (cum laude) in Electronic Engineering and the Ph.D. Degree in Electrical and Automation Engineering from the University “Mediterranea” of Reggio Calabria, Italy, in 2002 and 2006, respectively. Since 2005, he has been Postdoctoral Researcher of Electrical and Electronic Measurements at the Department of Information Engineering, Infrastructure and Sustainable Energy of the same University. At the present he is an Associate Professor and the Scientific Director of the Advanced Thermography Center at University Mediterranea. His main research interests include the design and characterization of distributed and intelligent measurement systems, advanced thermography, wireless sensor networks, environmental monitoring, decision-making problems and measurement uncertainty, process quality assurance, instrumentation reliability and calibration, energy, smart grids, battery testing, biomedical applications and statistical signal processing, noninvasive systems, biotechnologies and measurement, instrumentation, and methodologies related to healthcare.
Prof. Morello is a member of IEEE, he serves as AE for the IEEE Sensors J and he is member of Editorial Board of Scientific Reports, Nature Publishing Group.

Eulalia Balestrieri (Member, IEEE) received the M.S. degree in software engineering and the PhD degree in information engineering from the University of Sannio, Benevento, Italy, in 2003 and 2007, respectively. She is currently an Associate Professor in the Department of Engineering at the University of Sannio. She participated in several nationally and internationally financed projects as a Researcher with the University of Sannio. She is involved in research in the fields of analogue-to-digital and digital-to-analogue converters, remote educational laboratories distributed over a wide-area network, measurement instrumentation for digital telecommunication networks, biomedical instrumentation and measurements, and drone measurements. Dr. Balestrieri is a member of the IEEE Working Groups TC25 Subcommittee on Objective Blood Pressure Measurement for the Development of the New Standard IEEE-1721 “Standard for Objective Measurement of Systemic Arterial Blood Pressure in Humans,” the IEEE TC10 Analog-to-Digital Converter Subcommittee of the Waveform Generation, Measurement and Analysis Committee for the Development of the New Standards IEEE2414 “Standard for Jitter and Phase Noise,” and the IEEE 1658 “IEEE Standard for Terminology and Test Methods for Digital-to-Analog Converters” and the revisions of IEEE 1057 “IEEE Standard for Digitizing Waveform Recorders,” the IEEE 1241 “IEEE Standard for Terminology and Test Methods for Analog-to-Digital Converters,” and the IEEE 181, “IEEE Standard for Transitions, Pulses, and Related Waveforms.” She received the Certificate of Appreciation from the IEEE Standards Association “for outstanding contributions to the development of IEEE Standard 1241TM-2010 IEEE Standard for Terminology and Test Methods for Analogue-to-Digital Converters” and “for outstanding contributions to the development of IEEE Standard 2414 TM-2020 IEEE Standard for Jitter and Phase Noise.”

Silvestro Roatta was born in 1968, received the degree in Electrical Engineering from the Politecnico di Torino (1993) and the PhD in Physiology from the University of Torino (1997). He has been visiting researcher at the National Institute of Working Life, University of Umea, Sweden, and at the Center for Sensorimotor Interaction, University of Aalborg (Denmark). He is currently Associate Professor in Physiology and head of the Integrative Physiology Lab at the dept. of Neuroscience, University of Torino. His research interests include somato-vegetative interaction in muscle function and near infrared spectroscopy and ultrasound methodologies for the assessment of vascular reactivity and volume status. He is also interested in motor control and in new approaches to the development of brain-computer interfaces. He has published over 100 papers in international journals and is co-inventor of 7 patents. He is part of the academic spin-off VIPER (Politecnico di Torino) and serves as associate editor for Applied Sciences and Frontiers in Vascular Physiology.
Project Summary
The project FITI-PAIN has addressed the issue of pain assessment by aiming to overcome the limitations of currently available evaluation tools. Pain is a debilitating condition affecting about 20% of the adult population worldwide. It has important physical, psychological, and social consequences and can affect quality of life. Today, pain assessment mainly relies on patients’ self-reports describing their pain perception. Consequently, its evaluation is highly subjective. In specific cases, for example, when the patient is unable to communicate his/her sensation due to disability, the algologist assesses pain by observing the patient’s face or behaviour. For these reasons, the development of objective tools for pain assessment remains a major challenge.
This project has aimed to address the lack of objective pain assessment tools by proposing an instrumental technique. In detail, the Research Team has developed an innovative thermographic technique, named functional Infrared Thermal Imaging (fITI). This technique has been applied to study the pain mechanism in both healthy volunteers and patients with chronic pain. The technique enabled the definition of a preliminary parameter for quantitatively assessing pain sensation.
Although a large-scale clinical trial will be required before such a parameter can be considered a diagnostic tool, it can already be regarded as a promising biomarker of pain sensation.
The main achievements of the project include the development of two thermography-based prototypes and a mathematical model that converts thermal signal into perfusion signal in both the time and frequency domains. Pain was experimentally induced in healthy volunteers through standardized stimuli, while a high-performance thermal camera was used to observe the body areas affected by pain.
This study has identified a clear alteration in the perfusion signal and in its spectral components. Since, in the absence of suitable treatments, chronic pain can compromise the immune system and interfere with the ability to eat, concentrate, sleep, or interact with others, the possibility of assessing pain quantitatively could provide algologists a useful tool to expedite and to optimize therapeutic administration.
The experimental study carried out on the blood flow dynamics in the presence of pain has shown a direct alteration of the perfusion spectral components in specific low-frequency ranges. Perfusion components have been investigated in both healthy subjects and subjects with pain. Whereas microvascular perfusion is, at rest, characterized by periodical oscillations, the low frequency blood flow component has shown evident alterations at the moment at which the pain is evoked. In detail, the Cold Pressor Test and a Wartenberg pinwheel have been used as standardized tools to evoke pain in the hands of volunteers. Both hands have been observed for 20 minutes to analyse such oscillatory components of blood flow. Results show that the perfusion component in specific Regions Of Interest (ROIs) directly correlates with pain sensation. The wrist shows a greater sensitivity than the other hand regions. Blood flow components show noticeable alterations when the subject experiences high-intensity pain.
Overall, the experimental activities confirmed the effectiveness of the developed technique as a promising instrumental-based tool for pain assessment. The research team expects the proposed approach to have a significant scientific impact in both academic and clinical contexts, thanks to its innovative contribution to pain evaluation.
Project Objectives
This project has enabled the research team to make significant progress in analysing the pain mechanism. The main goal achieved has been the definition of an innovative measurement technique able to provide an objective evaluation of pain sensation by means of quantitative data. In addition, the definition of a standardized procedure and protocol make data repeatable and reproducible. The validation on healthy subjects has been carried out in laboratory-controlled conditions to ensure the clear correlation between the evoked pain and the parameters used to quantify it. Although the measurement scale and associated measurement unit have not been completely defined (described only in terms of methodology and prototype testing with volunteer participants), the perfusion spectral components have shown to be valid and effective parameters for assessing pain.
Objectives Achieved
The project activities have allowed us to investigate the role of blood flow components in pain mechanism. Experimental results have shown that these components can be considered promising biosignals and biomarkers for pain assessment. The fITI technique has been developed and tested on volunteers to assess objectively the pain evoked by means of two standardized stimuli (the Cold Pressor Test and the use of a Wartenberg pinwheel). Pain evaluation has been possible by analysing changes in the power of the perfusion spectral components. Changes have been observed during any stimulus application. Reference values have been defined by analysing the signal trend during the phase at rest.
This general objective has been achieved by means of the following sub-objectives:
1) The thermographic methodology, named functional Infrared Thermal Imaging (fITI), has been developed. In detail, the analysis relies on an indirect estimation of perfusion by correlating skin surface temperature with the underlying microvascular activity. To model this relationship, a modified version of Pennes’ bioheat equation was employed, enabling the characterization of heat transfer mechanisms from microvessels to the skin surface. The thermal model allows to convert the skin temperature into the underlying perfusion. A Morlet wavelet analysis of the perfusion signal allows for a spectral filtering of the blood flow components;
2) A standardized measurement procedure has been developed to guarantee reliability, repeatability and reproducibility of data;
3) A measurement uncertainty model has been defined to assess uncertainty, to determine the budget of its significant sources and to evaluate how this propagates into the microvascular perfusion evaluation;
4) Experimental activities enabled the validation of the skin thermal conduction model describing the thermal waves propagation from the microvessels toward the skin surface to characterize the mathematical relationship between surface skin temperature and inner blood flow signal;
5) The features of the perfusion spectral components have been characterized at rest and in the absence of neurological disorders in order to define the reference model and the thresholds of each component;
6) A protocol has been defined for evoking neuropathic pain in volunteers using reproducible and repeatable stimuli. Two stimuli have been considered: 1) the Cold Pressor Test; 2) the use of a Wartenberg pinwheel;
7) The spectral power has been defined as a parameter to evaluate the pain intensity according to the deviation of the spectral components of blood flow from their respective reference thresholds;
8) Specific colour scales have been defined to highlight pain intensity in spectrogram images using four classes: absent (black-dark blue), mild (blue-purple), moderate (red-orange), severe (yellow-white);
9) The methodology has been validated by controlled experiments in which the pain mechanism has been activated.
Outcomes
The main outcomes of the project are the two developed prototypes/demonstrators (the 2D fITI system and the 3D fITI system) and the algorithm developed in order to convert the temperature signal into the perfusion signal and to extract the perfusion spectral components. Figure 1 shows the first prototype. Figure 2 shows the second prototype, where a rotating platform allows the patient’s body to rotate around his/her own axis in front of the thermal camera.

Figure 1 – 2D fITI System prototype.

Figure 2 – 3D fITI System prototype.
Project Results
The results and outputs of the project are:
1) Design and development of the first demonstrator: the 2D automated fITI measurement system (Technology Readiness Level=6);
2) Design and development of the second demonstrator: the 3D fITI measurement system (TRL=6);
3) A pain assessment coloured scale reported in the scalogram of the perfusion signal;
4) Basis for future studies on pain trajectories as a new challenging research activity;
5) The mathematical model converting the skin temperature signal into the perfusion signal;
6) The Morlet Wavelet algorithm extracting the spectral components of the perfusion signal.
7) The Reference Model of the regular perfusion spectral components reporting the frequency ranges and the power ranges of each component for healthy subjects;
8) Description and comparative analysis of the autonomic patterns of response to acute exposure to painful and non-painful stressors (CPT, ES, MC), in relation to local cutaneous and muscular hemodynamic changes and to spectral changes of skin blood flow.
9) The measurement procedure and protocols used to evoke pain and to perform the FITI-PAIN technique;
10) The image database of the experimentation, which is accessible on demand;
11) The website and the publications.
In the following Figure 3, the thermographic image of the volunteer’s hands is shown. The thermographic record was for 20 minutes. Several ROIs have been investigated. In the fifteenth minute, the right forearm was stimulated by using the Wartenberg pinwheel. The reported pain sensation was 7 on a scale from 0 to 10. Figure 4 reports the spectrogram of the perfusion signal of the wrist of the right hand. It is possible to observe the response, highlighted in yellow, to the painful stimulus.
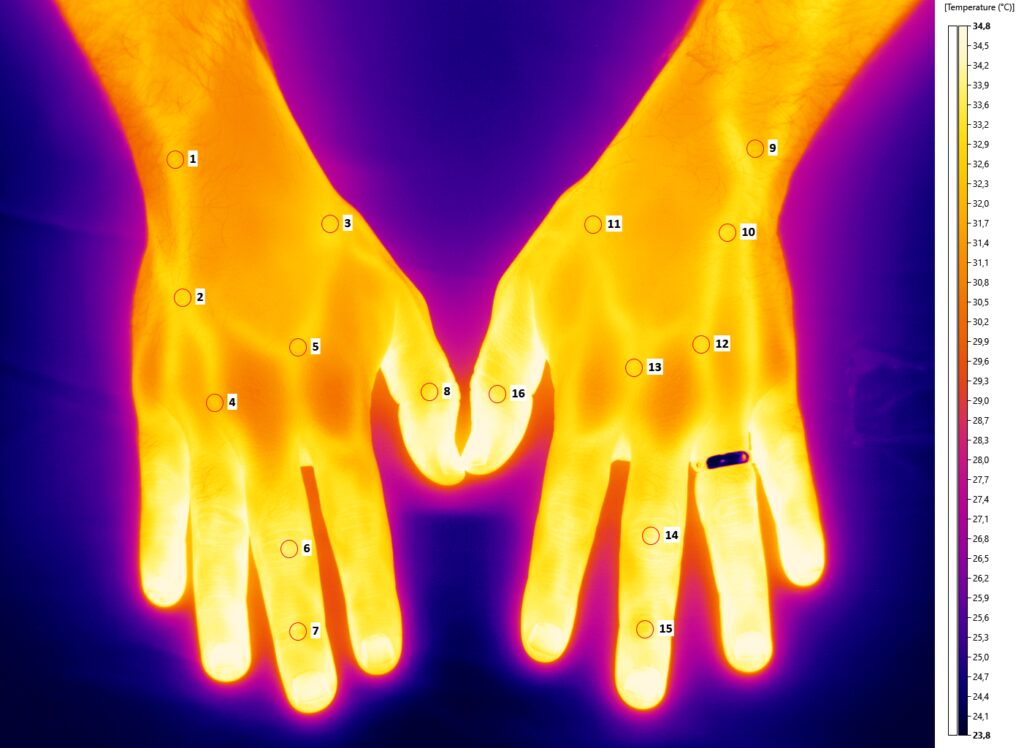
Figure 3 – Thermographic image of the volunteers’ hands.
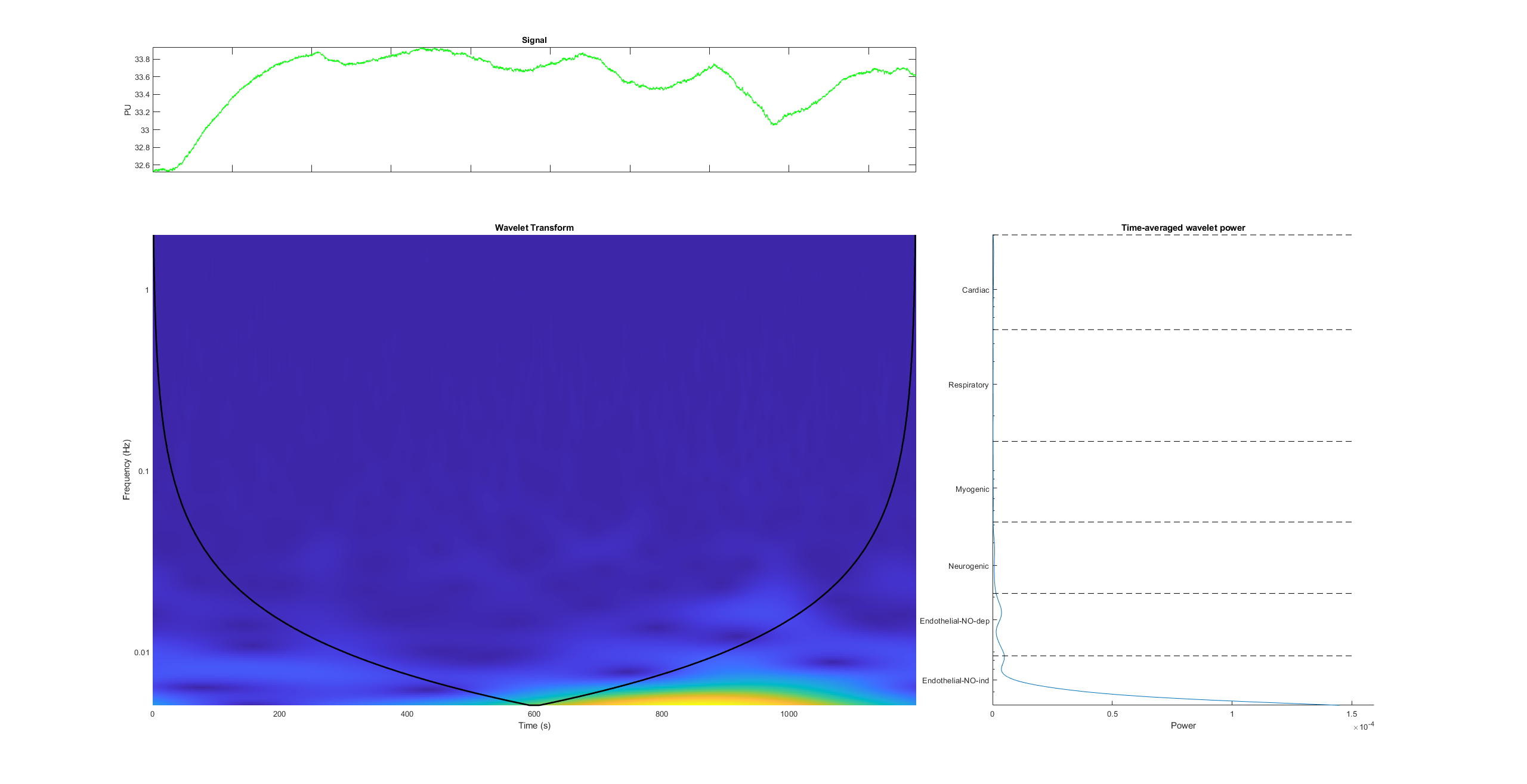
Figure 4 – Spectrogram of the response to pain of the right hand wrist (ROI #1).


